Investigator Profile: Raymond S.W. Yeung MD, FRCS (C), FACS
Pathogenesis of primary liver cancer
Current studies address the mechanisms of hepatic carcinogenesis focusing on the insulin-AKT-mTORC1 pathway. We use in vivo (genetic) models and cell-based assays to define the major 'drivers' involved in tumorigenesis. These studies have implications in human NASH-related liver disease and tumors.
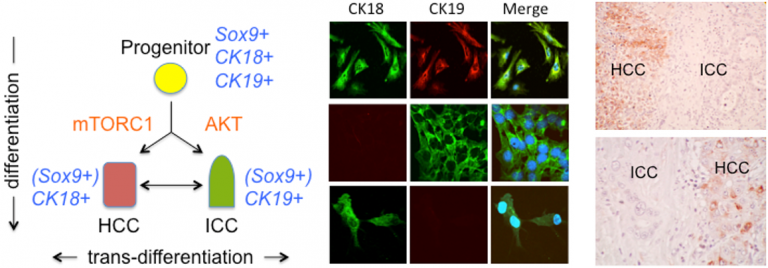
Figure 1. Role of Akt and mTORC1 in liver tumor differentiation. Bi-lineage progenitor cells (CK18+; CK19+) undergo differentiation to become hepatocellular (HCC) (CK18+) or cholangiocarcinoma (ICC) (CK19+) under the influence of mTORC1 or Akt, respectively. Immunohistochemistry showing an example of a human mixed HCC-ICC with differential expression of phospho-S6 (top panel) and phospho-Akt (bottom panel).
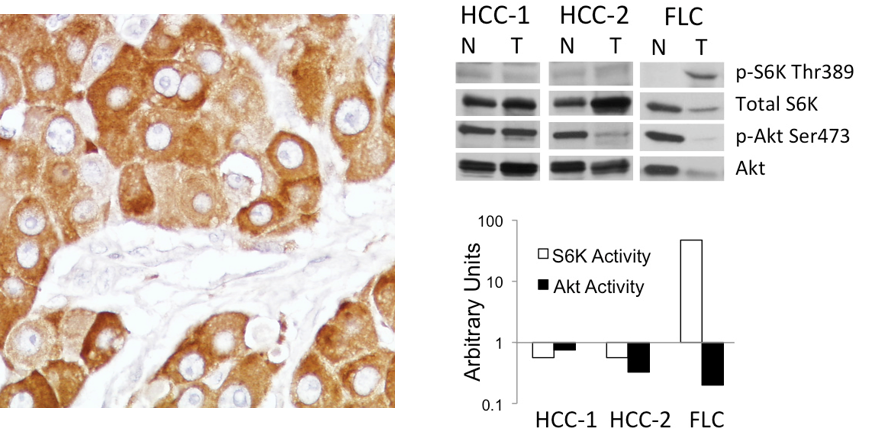
Figure 2. Right: mTORC1 activation in human fibrolamellar HCC (FLC). A) Immunohistochemical analysis of phospho-S6, an effector of mTORC1, showing strong reactivity in tumor cells. Left: mmunoblot analyses of Akt and S6K phosphorylation in FLC vs. classic HCC highlight differences in relative mTORC1 activity in the tumors.
Molecular carcinogenesis of fibrolamellar carcinoma (FLC)
In collaboration with Dr. Kimberly Riehle and scientists in the Department of Pharmacology, we are investigating the molecular mechanisms responsible for the pathogenesis of FLC, a rare form of HCC occurring in otherwise young, healthy population.
Biomarkers in hepatocellular carcinoma (HCC)
Clinically useful markers of HCC are very limited. As a crucial organ responsible for metabolism, liver and its associated tumors are expected to alter multiple metabolic pathways whose products are detectable in the serum. In collaboration with Dr. Daniel Raftery, Head, Metabolomic Lab, we are conducting studies to identify and validate serum metabolic markers that could potentially help detect HCC of various etiologies.
Molecular pathogenesis of tuberous sclerosis complex (TSC)
Although the underlying genetics are well established for TSC, the mechanisms through which alteration in the TSC/mTORC1 pathway affects cell growth and proliferation that lead to the spectrum of clinical disorders associated with TSC remains elusive. Our current studies investigate the role of mTORC1 on microtubule organization and function including protein trafficking.
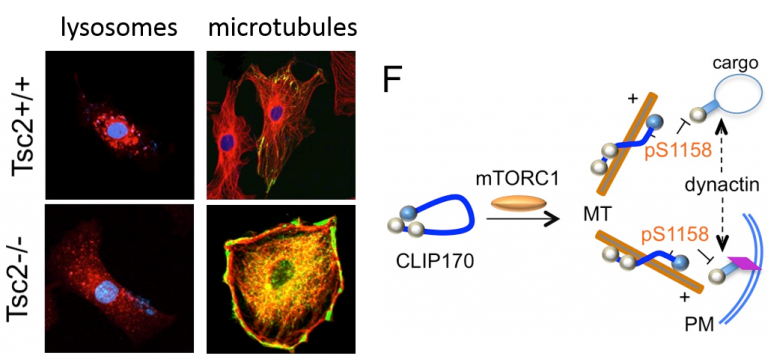
Figure 3. Constitutive activation of mTORC1 in the Tsc2-/- cells results in abnormal lysosome trafficking (highligthed by LysoTracker), and microtubule disorganization with bundles of MT (red) in the cell perimeter that localized with CLIP170 (green). Model shows the effects on mTORC1 on CLIP170 function in directing MT-dependent trafficking of cargo and cortical sensing of the plasma membrane (PM) during MT growth.


