Investigator Profile: Thomas S. Hatsukami, MD

Thomas S. Hatsukami, MD
Professor, Division of Vascular Surgery
Co-Director, Vascular Imaging Laboratory
V. Paul Gavora and Helen S. and John A. Schilling Endowed Chair in Vascular Surgery
Cardiovascular disease (CVD) is the number one cause of death worldwide and is a leading cause of long-term disability. It is estimated that the annual cost for the care of victims of CVD is over $390 billion per year in the U.S. alone. Most CVD events, such as heart attack and stroke, are atherosclerosis-related. Traditionally, the degree of vessel lumen narrowing has been used to identify the high-risk atherosclerotic plaque. However, there is increasing evidence that the structure, composition, and inflammatory activity of the atherosclerotic lesion are more important markers of the vulnerable plaque. Progress in understanding how vulnerable plaques develop has been hindered by our inability to serially examine these critical characteristics of the diseased vessel wall in a non-invasive fashion.
The mission of our research group is to advance high-resolution magnetic resonance imaging (MRI) technology for accurate, non-invasive examination of atherosclerosis. Our laboratory is organized along five core functions: 1) Imaging Physics: develop novel image acquisition techniques; 2) Histology: provide the histological gold-standard for validation of MRI findings; 3) Imaging Software: build custom-designed tools that permit more efficient, reproducible, quantitative image analysis; 4) Clinical Studies: apply MR imaging techniques to understand mechanisms leading to development of the vulnerable plaque; and 5) Reading Center: provide training, quality control, and image analysis for multi-center clinical trials using MRI.
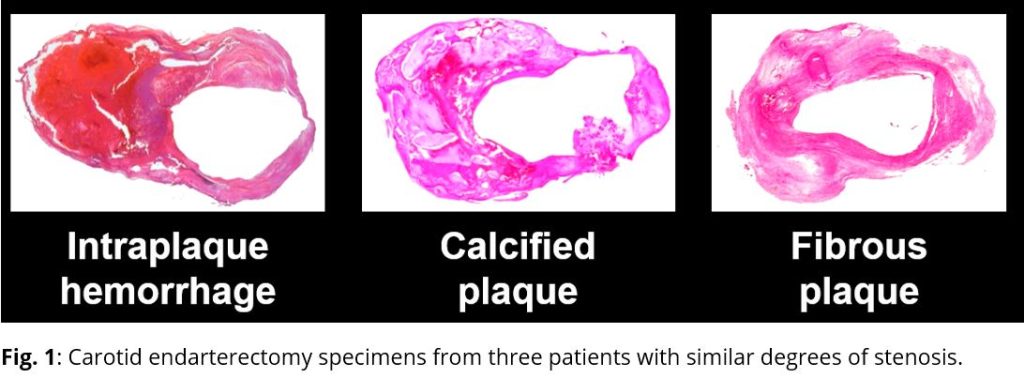
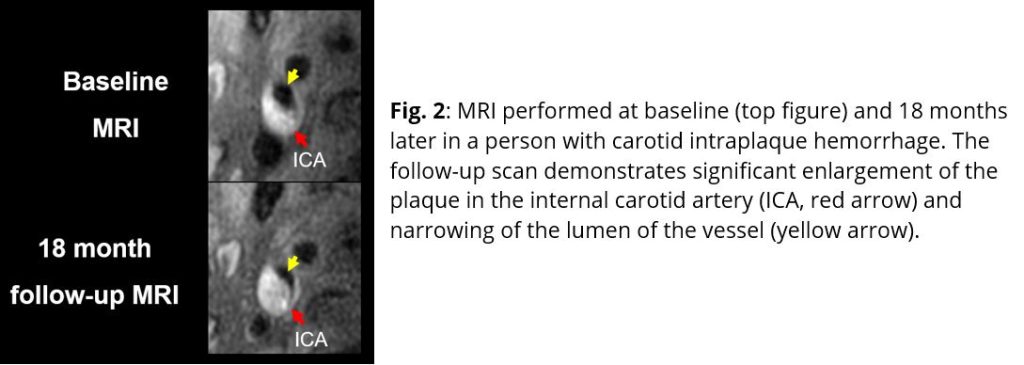
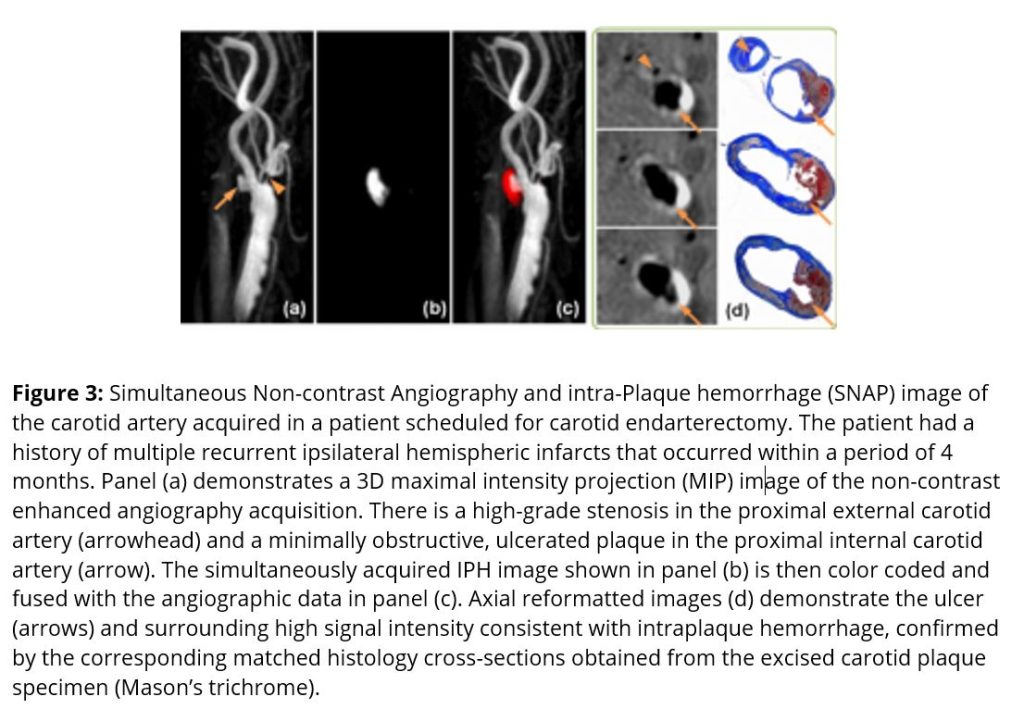
With funding from the National Institutes of Health, the National Science Foundation in China, and from industry, we have established a network of research centers using a standardized MRI protocol in North America, Japan, and China. Combined, these centers have enrolled over 2,000 individuals in prospective studies for which we serve as the Core Laboratory. Results from these studies have demonstrated that carotid plaques with MRI-detected intraplaque hemorrhage (IPH) are associated with more rapid progression, those with IPH and luminal surface disruption (Figure 1) are associated with a significantly higher risk for subsequent ipsilateral cerebrovascular ischemic events, and that treatment with aggressive lipid lowering therapy is associated with carotid plaque regression.
Increasingly, MRI is becoming accepted as an essential research tool for studying the pathophysiology of human atherosclerosis progression and regression, and for non-invasive identification of the high-risk plaque. A better understanding of disease mechanisms and factors leading to more rapid progression will permit identification of high-risk individuals for more aggressive treatment and potentially lead to the development of novel methods for therapeutic intervention.